- Blog
- Genogram example social work template
- Acer aspire keyboard driver download
- Positions in soccer numbers
- Does omnisphere 2 include omnisphere 1
- Density of water lbmft3
- Download google duo for pc windows 7
- Free svg betty boop
- Ds4 windows controller profile
- Breaking the quiet ep 3
- Work schedule monthly templates free
- Sight words for 1st grade pdf
- Nate newton pot
- Sony vegas pro serial number
- Attack on titan games free online
- Free schedule template pdf daily
- Coloring pages airplane
- Download photoshop cs6 master for mac torrent
- Google authenticator backup codes digits
- Ccleaner free download for windows 7
- Rpg maker mv 3d battle system
- Bears schedule print your brackets
- Mx player pc app free download
- Card games online free hearts
- Railway series collection
- Digital planner goodnotes free download
- Poster presentation size
- Gaming youtube banner
- Whatsapp windows desktop no more notification sound
- Mac laptop screen black extending desktop
- Blender donut model icing cuts problem
- Family genogram legend
- Windows 10 current lock screen image location
- Solitaire green felt klondike
- Car mechanic simulator 2018 game free download
- Internet connection stability test
- Weekly work schedule template free
- Best free solitaire download
- Pardes mein hai mera dil episode 9
- Emulator for xbox 360 rgh
- Minion butt plug
- Life coaching wheel of life worksheet
- Play bejeweled 3 online free popcap 2
- Blog
- Genogram example social work template
- Acer aspire keyboard driver download
- Positions in soccer numbers
- Does omnisphere 2 include omnisphere 1
- Density of water lbmft3
- Download google duo for pc windows 7
- Free svg betty boop
- Ds4 windows controller profile
- Breaking the quiet ep 3
- Work schedule monthly templates free
- Sight words for 1st grade pdf
- Nate newton pot
- Sony vegas pro serial number
- Attack on titan games free online
- Free schedule template pdf daily
- Coloring pages airplane
- Download photoshop cs6 master for mac torrent
- Google authenticator backup codes digits
- Ccleaner free download for windows 7
- Rpg maker mv 3d battle system
- Bears schedule print your brackets
- Mx player pc app free download
- Card games online free hearts
- Railway series collection
- Digital planner goodnotes free download
- Poster presentation size
- Gaming youtube banner
- Whatsapp windows desktop no more notification sound
- Mac laptop screen black extending desktop
- Blender donut model icing cuts problem
- Family genogram legend
- Windows 10 current lock screen image location
- Solitaire green felt klondike
- Car mechanic simulator 2018 game free download
- Internet connection stability test
- Weekly work schedule template free
- Best free solitaire download
- Pardes mein hai mera dil episode 9
- Emulator for xbox 360 rgh
- Minion butt plug
- Life coaching wheel of life worksheet
- Play bejeweled 3 online free popcap 2
Pharmaceuticals: The specific gravity is used to find out the purity of a drug since each of the constituents has a distinct specific gravity.Specific Gravity has a wide range of applications including: Other instruments to measure specific gravity are the Pycnometer, and digital density meters based on oscillating U-tubes.
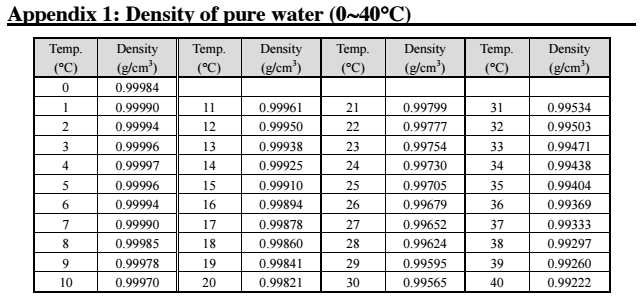
The device is designed to float freely at the liquid surface with a protruding stem giving a reading corresponding to the specific gravity of the liquid. The hydrometer can be used to measure the specific gravity of any liquid. There is a wide range of instruments designed to measure the specific gravity of a material. The reference density of water at 4 oC (39 oF) is used as the reference as these are the conditions of maximum density. Where SG = specific gravity, ρ = density of the material (kg/m 3), ρW = density of water ( kg/m 3). The following formula is used to calculate the specific gravity of a material. The specific gravity of all other materials is compared to water as a fraction heavier lighter or heavier density. The specific gravities of gases usually are compared to dry air which generally has a density of 1.29 g per litre.

By definition, water has a density of 1 kg per litre at this temperature. In more general terms specific gravity is the ratio of the density of a material to that of any standard substance, although usually this is water at 4 degrees Celsius or 39.2 degrees Fahrenheit. Specific gravity is unique to every material and has a very wide range of application. The temperature and pressure of both the material and water need to be the same as these factors influence the density and hence the specific gravity. The Specific Gravity of liquids and solids is defined as a dimensionless unit which is the ratio of density of a material to the density of water at a given temperature, where density is defined as the material’s mass per unit volume and is measured in kg/m 3.
